IONIC & COVALENT BONDS
Ionic Bonds
Ionic bonding involves the complete transfer of valence electrons between atoms, resulting in the formation of two oppositely charged ions. Metals lose electrons to become positively charged cations, while nonmetals accept those electrons to become negatively charged anions. Ionic bonds require a metal as an electron donor and a nonmetal as an electron acceptor.
Ionic bonding occurs because metals typically have few electrons in their outermost orbitals. By losing these electrons, metals can achieve a stable noble gas configuration and fulfill the octet rule. In ionic bonding, multiple electrons can be donated or received to satisfy the octet rule. The charges on the resulting anion and cation depend on the number of electrons donated or received. In an ionic compound, the sum of the charges must be zero to maintain overall electrical neutrality.
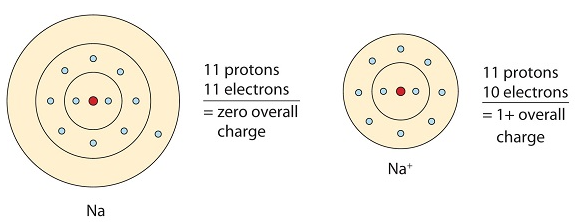
This sodium molecule donates the lone electron in its valence orbital in order to achieve octet configuration. This creates a positively charged cation due to the loss of electron.
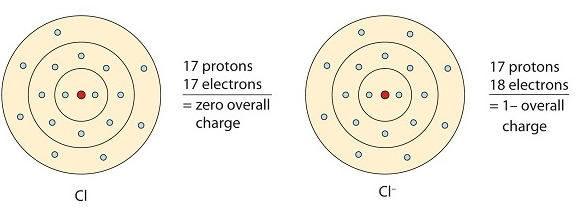
This chlorine atom receives one electron to achieve its octet configuration, which creates a negatively charged anion
Note:
The predicted overall energy of the ionic bonding process, which includes the ionization energy of the metal and electron affinity of the nonmetal, is usually positive, indicating that the reaction is endothermic and unfavorable. However, this reaction is highly favorable because of the electrostatic attraction between the particles. At the ideal interatomic distance, attraction between these particles releases enough energy to facilitate the reaction. Most ionic compounds tend to dissociate in polar solvents because they are often polar. This phenomenon is due to the opposite charges on each ion.